What We Accomplished in 2021-2022
Network Recruitment
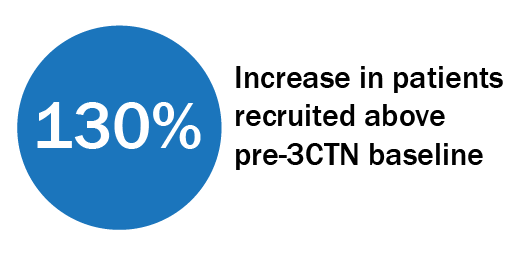

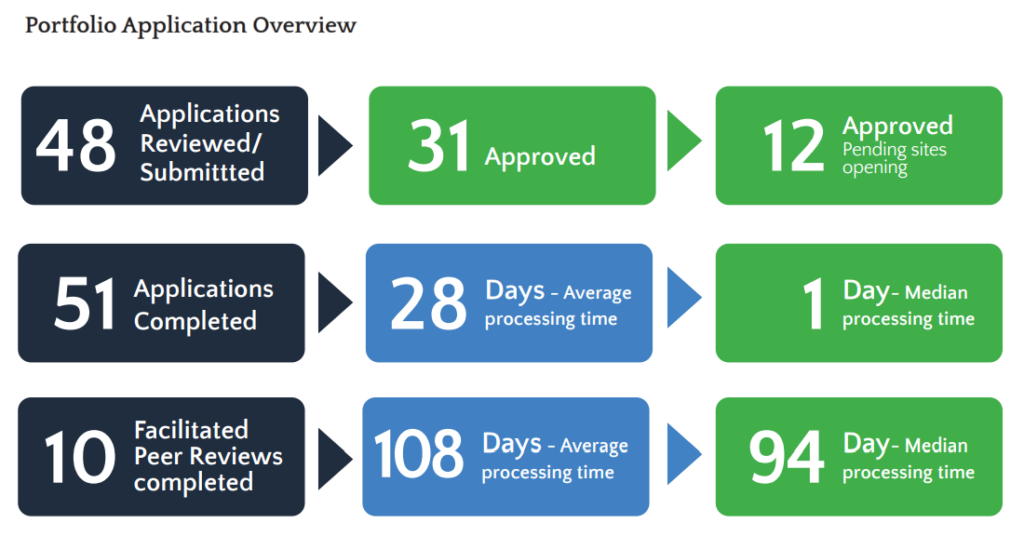
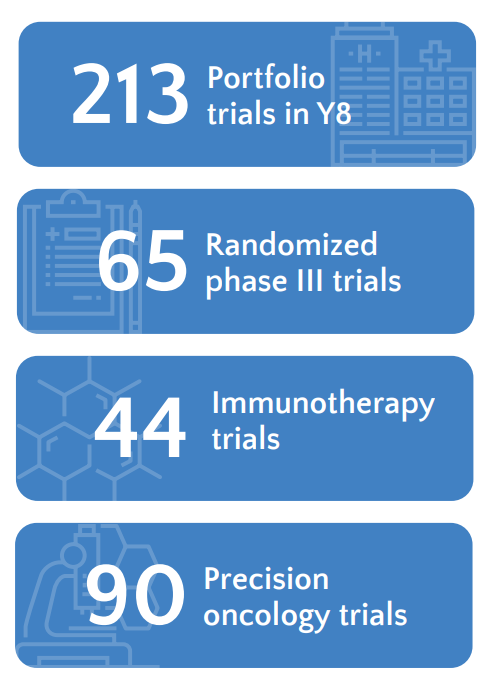
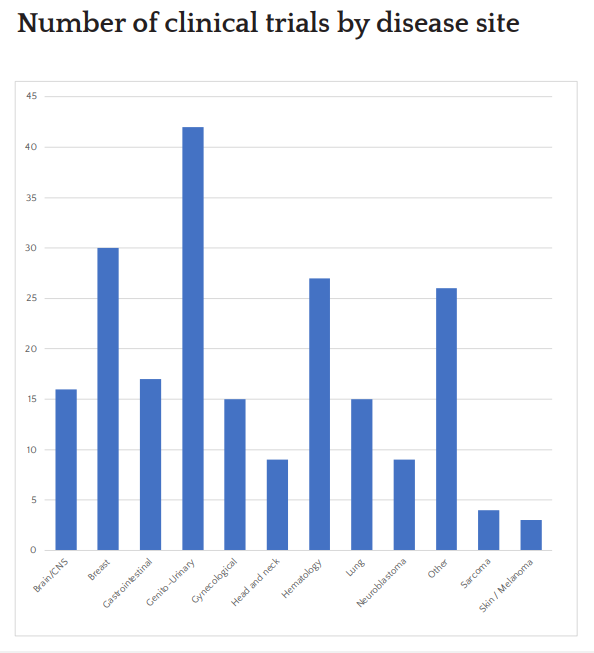
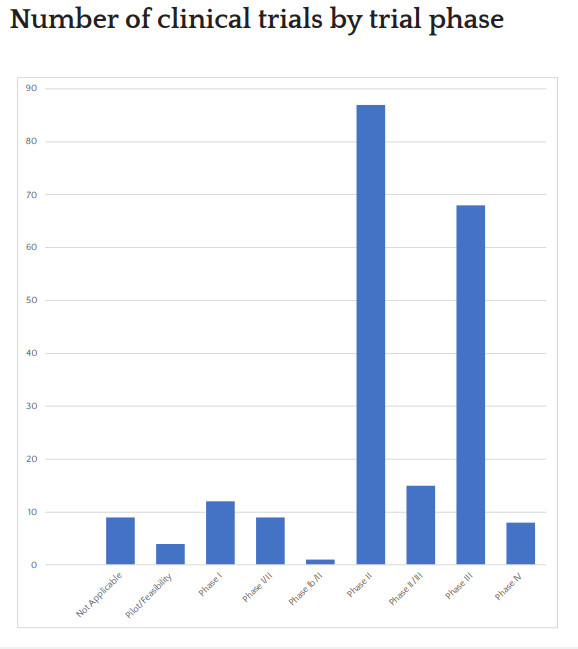
Trial Portfolio
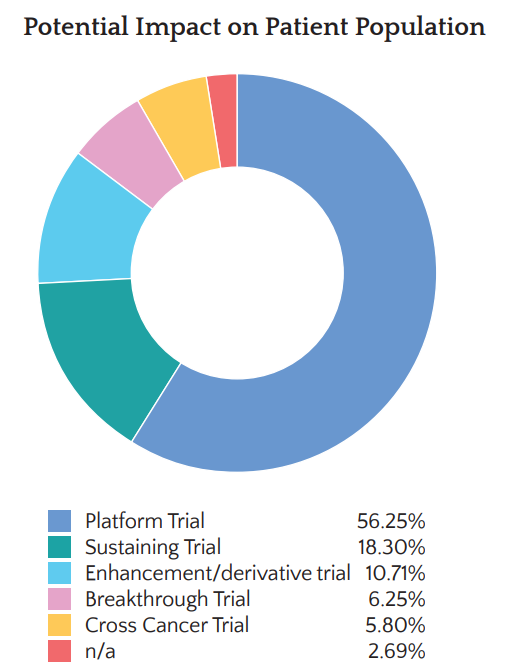
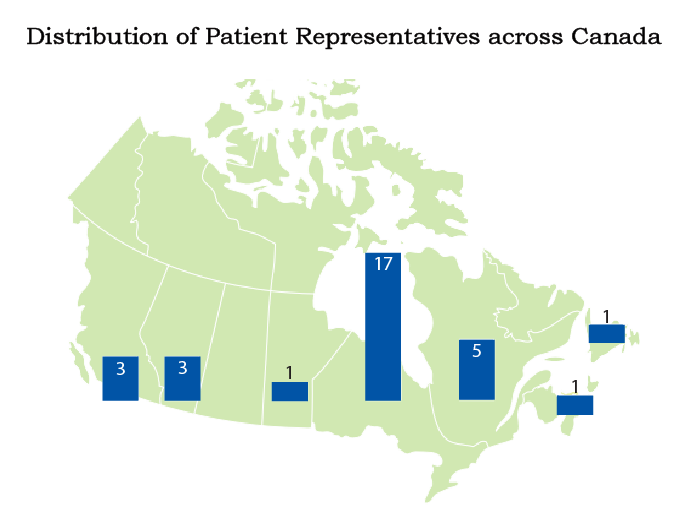
Patient and Public Involvement
Performance by Province
Alberta
Accrual to target 91% | 3 Patient partners
- Implemented a new Clinical Trials Management System (CTMS) that will result in improved trial efficiency.
- Engaged Patient Advisors in the implementation of a new electronic medical record (EMR) system. Patients can now be contacted via MyChart for potentially eligible trials.
- Training courses to address biosafety in clinical trials for staff members is under development.
British Columbia
Accrual to target 84% | 3 Patient partners
- Linked the new clinical trials website to the CTMS to provide an updated list of open trials for patients.
- Implemented a new Phase 1 room to allow patients on Phase 1 clinical trials to rest post-treatment.
Manitoba
Accrual to target 34% | 1 Patient partner
- Launched a new business unit model to allow greater physician engagement and more access to trials for patients.
- Held daily virtual events during International Clinical Trials Week and engaged Patient Representative to participate in events.
Newfoundland
Accrual to target 38% | 1 Patient partner
- Launched and implemented Clinical Trials Management System – EDGE.
- Listed all open trials on Eastern Health website for patients and stakeholders.
Nova Scotia
1 Patient partner
- Developed a Clinical Trials Working Group (CTWG) to address trial start-up, feasibility and patient population for recruitment.
- Added a Patient Representative to the CTWG to provide feedback on potential upcoming trials
Ontario
Accrual to target 151% | 17 Patient partners
- Developed clinical trial resources to support patients, families and friends.
- Implemented initiatives to improve trial selection and trial activation processes.
- Developed quality improvement initiatives to standardize processes and empower individuals to solve problems and integrate quality improvement into day-to-day work.
- Collaborated with other Network sites on best practices for audits.
Quebec
Accrual to target 193% | 5 Patient partners
- Developed and launched Clinical Trials Navigator pilot project, a first of its kind in the province.
- Redesigned and relaunched a new version of OncoQuébec, an online resource for sourcing available cancer clinical trials within the province of Quebec, based on user feedback.
Pediatrics
14 Patient partners
- C17 launched and promoted U-Link, a resource to improve access to clinical trials among the pediatric cancer sites.
- Three High Priority Trials (HPT) were identified and prioritized for trial activation. 94 per cent of participating sites activated HPTs within 90 days.

See Our Annual Report
Get highlights of the network’s achievements and learn about our goals and strategy moving forward.
